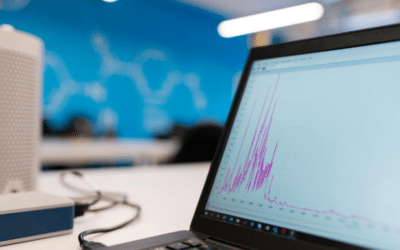
Improvement of Peak Integration in Capillary Electrophoresis: Reference Data Set No. 1
AdvertisementIntroduction As biopharmaceuticals grow in complexity, traditional peak integration algorithms are increasingly struggling to keep pace, particularly in Capillary Electrophoresis (CE), where intricate sample profiles expose the limits of conventional...
Peak Integration of Electropherograms in GMP and Research Labs: Navigating Increased Scrutiny Amid Data Integrity Audits and Inspections
AdvertisementIntroduction Capillary electrophoresis (CE) offers superior separation for complex biologics like monoclonal antibodies, but its irregular baselines and broad, asymmetric peaks make peak integration far more challenging than chromatography—especially in...
Separation of virus-like particles and nano-emulsions for vaccine development by Capillary Zone Electrophoresis
AdvertisementIntroduction Vaccine formulations increasingly incorporate virus-like particles (VLPs) along with nano-emulsion adjuvants to boost immune responses. However, analytically characterising these mixtures is very difficult because both components are...
A Comprehensive Evaluation of Analytical Method Parameters Critical to the Reliable Assessment of Therapeutic mRNA Integrity by Capillary Gel Electrophoresis
AdvertisementIntroduction Messenger RNA (mRNA)-lipid nanoparticle (LNP) therapies are an innovative biopharmaceutical approach with quickly adaptable production methods. However, evaluating their quality requires specific analytical techniques that consider the unique...
Universal Study Design for Instrument Changes in Pharmaceutical Release Analytics
AdvertisementIntroduction Introduces a universal study design to assess comparability when switching or updating pharmaceutical analytical instruments, ensuring compliance with GMP requirements. Demonstrates the approach with a benchmark study comparing ICE3 and...
Advancing Reversed-Phase Chromatography Analytics of Influenza Vaccines Using Machine Learning Approaches on a Diverse Range of Antigens and Formulations
AdvertisementIntroduction Reversed-phase chromatography is a vital tool in influenza vaccine analysis, yet optimising methods for difficult separations - like strain co-elution or excipient interference - has traditionally depended on manual interpretation and analyst...
ICH Q14: Guideline Overview for Analytical Procedure Lifecycle Management
AdvertisementThe ICH Q14 guideline, formally part of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH), represents a step forward in ensuring that analytical methods for pharmaceuticals are scientifically...
A Practical Approach to Implementing ICH Q14: Tools for Analytical Quality by Design in Capillary Electrophoresis Method Development
AdvertisementIntroduction Practical, stepwise implementation of ICH Q14 using AQbD tools Full method lifecycle approach: from Analytical Target Profile (ATP) and technology selection to control strategy and lifecycle management Efficient application of Design of...
Advertisement
Advertisement
Advertisement